Back to article: Evolution of substrate specificity in the Nucleobase-Ascorbate Transporter (NAT) protein family
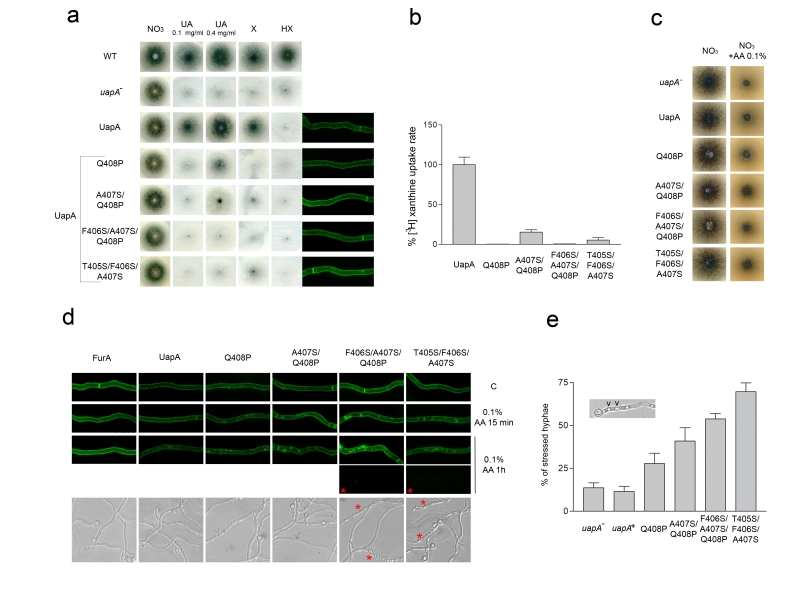
FIGURE 2: Specific residues within the NAT signature motif are critical for UapA function and specificity. (a) Growth tests, on standard MM, of isogenic strains expressing wild-type (UapA) or mutant UapA versions (Q408P, A407S/Q408P, T405S/F406S/A407S and F406S/A407S/Q408P), expressed in a genetic background genetically lacking other major purine transporters (i.e. ΔuapC ΔazgA). A strain lacking a functional UapA (uapA–) and a standard wild-type A. nidulans strain (WT) are also included in the test as controls. The test depicts growth on 10mM sodium nitrate (NO3) or 0.1 mg/ml uric acid (UA), xanthine (X), hypoxanthine (HX), after 48 h at 37°C. A 4-fold higher concentration of UA (0.4 mg/ml) was also used in order to distinguish UapA mutants with highly reduced affinity for substrates (Q408P, A407S/Q408P) from mutants with an apparent total loss of transport activity (T405S/F406S/A407S and F406S/A407S/Q408P). Subcellular localization of wild-type or mutant UapA-GFP versions, as revealed by in vivo epifluorescence microscopy, is also shown at the right side of the panel. Notice that in all cases UapA-GFP is stably localized in the plasma membrane and the septa of A. nidulans hyphae. For details of sample preparation see Material and methods. (b) Comparative UapA-mediated uptake rates of radiolabeled 3H-xanthine in strains expressing wild-type (UapA) or mutant versions (Q408P, A407S/Q408P, T405S/F406S/A407S and F406S/A407S/Q408P) of UapA-GFP. Results are averages of three measurements for each concentration point. SD was < 20%. (c) Growth tests, at 37°C, on standard MM supplemented with 10 mM sodium nitrate (NO3) as nitrogen source in the absence or presence of 0.1% L-ascorbate (AA) of isogenic strains lacking UapA (uapA–), or expressing wild-type (UapA) or mutant UapA versions (Q408P, A407S/Q408P, T405S/F406S/A407S and F406S/A407S/Q408P). (d) Upper panel: In vivo epifluorescence microscopy following the effect of L-ascorbate (0.1% AA) on UapA-GFP or FurA-GFP subcellular localization. Notice that after 15 min addition of AA clear endocytic turnover of UapA-GFP (i.e. appearance of cytoplasmic structures corresponding to vacuoles; Gournas et al., 2010) is observed in all UapA mutants, but not in wild-type UapA or FurA. Vacuolar turnover is more evident after 1 h of AA in the triple mutants T405S/F406S/A407S and especially F406S/A407S/Q408P, where more and larger vacuoles appear. Notice that after 1 h of AA, moderate vacuolar turnover is also evident in the wild-type UapA, but not in FurA. Noticeably also, 1 h in AA triggers “loss” of GFP fluorescence in a significant fraction (> 50%) of hyphae in in the triple mutants T405S/F406S/A407S and F406S/A407S/Q408P (lowest black boxes marked with a red asterisk). The same effect, albeit significantly reduced, is also seen in A407S/Q408P (36%) and Q408P (22%), while in the wild-type UapA-GFP or FurA-GFP fluorescence loss is < 10% (not shown). Lower panel: L-ascorbate (1% AA for 1 h) triggers differential hypervacuolarization and reduction of hyphal width (i.e. cell stress) in UapA mutants and controls. Stressed hyphae are marked with a red asterisk. (e) Quantification of the effect of L-ascorbate on hyphal morphology, as recorded for 100 hyphae, of each strain analyzed. In all cases, hypervacuolarization and reduction of the width of hyphae was associated with loss of fluorescence (not shown). The results depicted in the graph confirm the apparent cytotoxicity of L-ascorbate in the triple mutants T405S/F406S/A407S and F406S/A407S/Q408P, where 50-70% of hyphae seem stressed, followed by progressively lower effects on A407S/Q408P (40%), Q408P (27%) and wt UapA (11%).