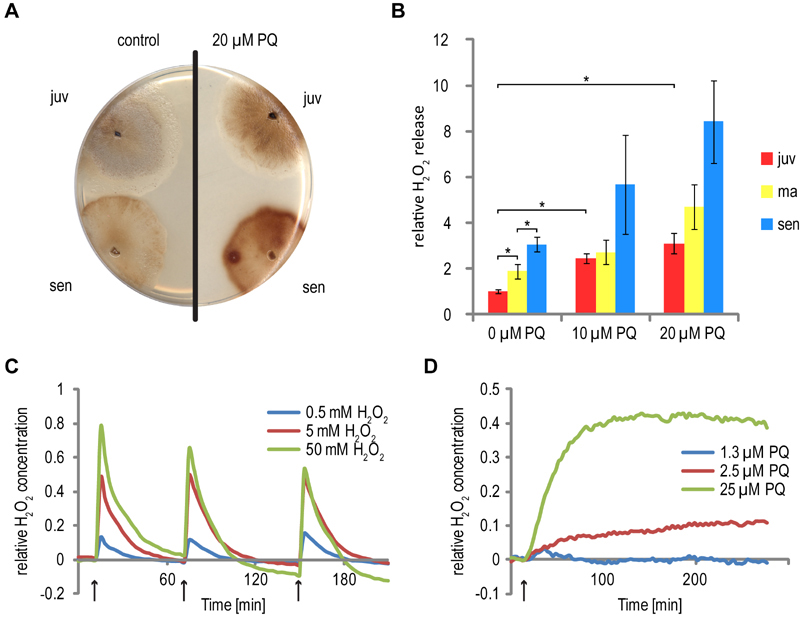
FIGURE 1: Increased hydrogen peroxide generation and release by P. anserina cultures during aging and PQ-stress.
(A) After 2 days of ascospore germination, fungal mycelia were either directly challenged with 20 µM PQ (juv) or grown for 11 days on M2 medium to obtain a senescent stage. The cultures were subsequently incubated for 4 days on M2 with 20 µM PQ. As a control, both age stages (juv: 6 days; sen: 17 days) were cultivated on PQ-free M2 medium. After this time, DAB-staining-solution was poured on the cultures. The reaction with secreted H2O2 leads to the formation of a dark brown pigment.
(B) Quantitative measurement of H2O2 release. P. anserina cultures were essentially grown as described in (A). Instead of on M2, cultures were grown on PASM medium until reaching a final age of 6 days (juv), 11 days (ma) and 15 days (sen), respectively. Concentrations of 0 µM, 10 µM and 20 µM PQ were administered. Bars indicate mean ± standard error of H2O2 release relative to juvenile untreated cultures. n = 16 – 41. Asterisks indicate pV < 0.01.
(C and D) Relative changes in H2O2 concentration in the cytoplasm. P. anserina strains producing the redox sensitive HyPer protein, localized to the cytoplasm, were germinated for 2 days on germination medium and grown for 4 days on M2 agar plates. The mycelium was punched out of the agar, transferred to a 96 well plate and soaked in 60 µl water. Subsequently, H2O2 (C) or PQ (D) was added at the indicated times (arrows) to a final concentration of 0.5 mM, 5 mM and 50 mM or 1.3 µM, 2.5 µM and 25 µM, respectively. The fluorescence of the mycelium after exitation at 488 ± 5 nm (oxidized HyPer) and 420 ± 5 nm (reduced HyPer) was measured at 530 ± 20 nm. The graphs show the ratio of oxidized to reduced HyPer normalized to a untreated HyPer strain.