Research Reports:
Microbial Cell, Vol. 9, No. 6, pp. 126 - 132; doi: 10.15698/mic2022.06.778
Investigating the role of G-quadruplexes at Saccharomyces cerevisiae telomeres
1European Research Institute for the Biology of Ageing, University of Groningen, University Medical Center Groningen, Groningen, The Netherlands
Keywords: telomere, G-quadruplex, budding yeast, tlc1-tm, Cdc13, Pif1, Saccharomycotina.
Received originally: 12/04/2022 Received in revised form: 09/05/2022
Accepted: 16/05/2022
Published: 19/05/2022
Correspondence:
Michael Chang, European Research Institute for the Biology of Ageing, University of Groningen, University Medical Center Groningen, Groningen, The Netherlands m.chang@umcg.nl
Conflict of interest statement: The authors declare no conflict of interest.
Please cite this article as: Sonia Stinus, Fernando R. Rosas Bringas, Lisa Wanders, and Michael Chang (2022). Investigating the role of G-quadruplexes at Saccharomyces cerevisiae telomeres. Microbial Cell: 9(6):126-132. doi: 10.15698/mic2022.06.778
Abstract
The G-quadruplex consensus motif G≥3NxG≥3NxG≥3NxG≥3 is found at telomeres of many species, ranging from yeast to plants to humans, but the biological significance of this fact remains largely unknown. In this study, we examine the in vivo relevance of telomeric G-quadruplexes in the budding yeast Saccharomyces cerevisiae by expressing a mutant telomerase RNA subunit (tlc1-tm) that introduces mutant [(TG)0–4TGG]xATTTGG telomeric repeats instead of wild-type (TG)0-6TGGGTGTG(G)0-1 repeats to the distal ends of telomeres. The tlc1-tm telomere sequences lack the GGG motif present in every wild-type repeat and, therefore, are expected to be impaired in the formation of G-quadruplexes. Circular dichroism analysis of oligonucleotides consisting of tlc1-tm telomeric sequence is consistent with this hypothesis. We have previously shown that tlc1-tm cells grow similarly to wild-type cells, suggesting that the ability to form telomeric G-quadruplexes is not essential for telomere capping in S. cerevisiae cells.
INTRODUCTION
The physical ends of eukaryotic chromosomes are protected by nucleoprotein complexes known as telomeres. Telomeres protect chromosome ends from degradation, from telomere-telomere fusion events, and from being recognized as double-stranded DNA breaks [1]. In most eukaryotic species, telomeres consist of double-stranded G/C-rich DNA followed by a G-rich 3′ single-stranded overhang. Proper telomere function is ensured by the specialized proteins bound to the double-stranded and single-stranded telomeric repeats. Telomere length is kept in a state of dynamic equilibrium. Incomplete DNA replication and nucleolytic degradation cause telomeres to shorten, while the reverse transcriptase telomerase is responsible for telomere lengthening [1]. Telomerase extends the 3′ overhang of telomeres by iterative reverse transcription using its RNA subunit as a template.
–
Due to the G-rich nature of the telomeric repeats, telomeric DNA has the potential to form G-quadruplexes, which are highly stable secondary structures composed of Hoogsteen hydrogen-bonded guanines arranged in planar G-tetrads stacked together [2]. Intramolecular G-quadruplexes are predicted to form within sequences containing four runs of at least three guanines (G≥3NxG≥3NxG≥3NxG≥3), and the telomeric DNA of most eukaryotic organisms conform to this consensus sequence. While most studies on G-quadruplexes have been carried out in vitro, there is also in vivo work supporting the existence of G-quadruplexes at telomeres. The most direct evidence comes from studies in ciliates. The telomere-binding protein TEBPβ, from the related ciliates Oxytricha nova and Stylonychia lemnae, can promote the formation of G-quadruplexes in vitro [3][4]. Knockdown of TEBPβ in S. lemnae eliminates detection of telomeric G-quadruplexes in vivo using the Sty3 G-quadruplex antibody in nuclear staining experiments [4]. Telomeric G-quadruplexes are not detected during S phase, presumably to allow replication of telomeres [4]. Unfolding of telomeric G-quadruplexes during S phase requires phosphorylation of TEBPβ, as well as telomerase and a RecQ-like helicase [4][5][6].
–
In the budding yeast Saccharomyces cerevisiae, the main telomere binding protein Rap1, like TEBPβ, can bind and promote the formation of G-quadruplexes in vitro [7][8]. In contrast to the findings in ciliates, chromatin immunoprecipitation experiments using the BG4 G-quadruplex antibody suggest that telomeric G-quadruplexes may form in late S phase, when S. cerevisiae 3′ overhangs reach their longest length [9]. The telomerase subunit Est1 can also promote G-quadruplex formation in vitro, and cells expressing Est1 mutants deficient in this activity exhibit gradual telomere shortening and replicative senescence, suggesting a potential positive role for G-quadruplexes in telomerase-mediated extension of telomeres [10]. In addition, there is evidence to suggest that stabilization of G-quadruplexes suppresses the temperature sensitivity of the telomere capping-defective cdc13-1 mutant [11]. Cdc13 is a single-stranded telomeric DNA binding protein; the cdc13-1 mutant loses the ability to block excessive nucleolytic resection of telomeric DNA at elevated temperatures, resulting in an accumulation of single-stranded telomeric DNA [12][13]. The folding of this DNA into G-quadruplexes has been proposed to facilitate telomere capping by inhibiting further nucleolytic resection [11]. Despite these findings, it remains unclear whether G-quadruplexes have an evolutionarily conserved function in telomere biology [14].
–
In this study, we examined the function of G-quadruplexes at S. cerevisiae telomeres by expressing a mutant telomerase RNA subunit (tlc1-tm) that introduces [(TG)0–4TGG]xATTTGG mutant telomeric repeats instead of wild-type (TG)0-6TGGGTGTG(G)0-1 repeats [15][16]. The mutant repeats are impaired in the formation of G-quadruplexes, and we have previously shown that tlc1-tm repeats are poorly bound by Rap1 [17]. Despite being deficient in telomeric G-quadruplex formation, tlc1-tm cells are viable and grow as well as wild-type cells, suggesting that the ability to form telomeric G-quadruplexes is not essential for telomere capping and cell viability in S. cerevisiae.
RESULTS
tlc1-tm mutant telomere sequences have reduced potential to form G-quadruplexes
To assess the role of G-quadruplexes at yeast telomeres, we require a yeast strain with telomeric DNA sequences that lack the potential to form G-quadruplexes. Such a strain can be obtained by mutating the template sequence of the RNA subunit of telomerase, TLC1. The vast majority of mutations to the TLC1 template sequence causes disruption of telomerase enzymatic activity, and consequently, replicative senescence [18]. Those that do not are often associated with slow growth, dramatic alterations in telomere profile (i.e. elongated, very short, or extensively degraded), and aberrant chromosome separation and segregation [18][19]. The tlc1-tm mutant introduces [(TG)0–4TGG]xATTTGG mutant telomeric repeats instead of wild-type (TG)0-6TGGGTGTG(G)0–1 repeats, and grows similar to a wild-type strain, even when one telomere consists entirely of mutant sequence [15][16][17]. Telomeres in the tlc1-tm mutant are on average longer and more heterogeneous in length than in wild-type strains [17], but the telomere profile of tlc1-tm is much less dramatically altered compared to most other mutants of TLC1 with altered template sequences [18][19].
–
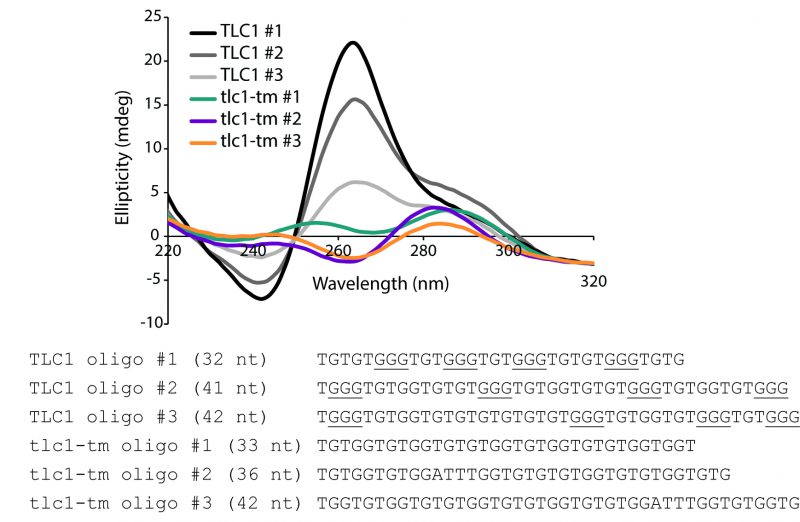
The lack of the GGG motif in the mutant repeat sequence should weaken the potential of G-quadruplex formation. To test this idea, we used the G-quadruplex prediction tool, G4Hunter, where a score greater than 1.2 indicates high G-quadruplex-forming potential [20]. While analysis of wild-type sequences gave G4Hunter scores of 1.366, 1.375, and 1.286 (see sequences used in Figure 1), none of the three analyzed mutant tlc1-tm sequences has a score greater than 1, thus indicating that the mutant telomeric sequences have reduced G-quadruplex-forming potential. To validate this hypothesis, we subjected oligonucleotides with either wild-type or tlc1-tm telomere sequences to circular dichroism (CD) analysis after incubation with potassium. In agreement with previous studies reporting that yeast telomeric DNA can fold into G-quadruplex structures in vitro [7][21], we find that all three oligonucleotides composed of wild-type telomeric sequence generate a negative peak at 240 nm and a positive peak at 263 nm (Figure 1), which is a pattern consistent with parallel G-quadruplex formation. In contrast, none of the oligonucleotides with tlc1-tm telomere sequence form such a pattern (Figure 1). It is formally possible that tlc1-tm telomeres form less stable two-quartet G-quadruplexes (which have a consensus sequence of G≥2NxG≥2NxG≥2NxG≥2). Indeed, the spectra of tlc1-tm oligonucleotides #2 and #3, despite having low amplitude, could indicate an antiparallel G-quadruplex structure, which is characterized by a negative peak near 260 nm and positive ones at 240 and 295 nm. Nevertheless, our findings indicate that the formation of any G-quadruplex structures by wild-type telomeric sequence should be, at minimum, greatly perturbed in tlc1-tm telomeric sequence.
Deletion of PIF1 suppresses cdc13-1, but not cdc13-1 tlc1-tm, temperature sensitivity
To test whether tlc1-tm telomere sequences are defective in forming G-quadruplexes in vivo, we stabilized G-quadruplexes in the telomere capping-defective cdc13-1 mutant by deleting PIF1. Pif1 is a helicase and a potent unwinder of G-quadruplexes [22]. Suppression of cdc13-1 temperature sensitivity by pif1Δ has already been reported [23]. We find that pif1Δ cannot suppress the temperature sensitivity of cdc13-1 in a tlc1-tm background (Figure 2A). We observe the same effect when using the pif1-m2 allele, which is specifically deficient for the nuclear isoform of Pif1 [24]. Thus, tlc1-tm telomeres remain uncapped even in the absence of Pif1, possibly due to a lack of G-quadruplexes to stabilize.
–
We noticed that cdc13-1 tlc1-tm cells grow more slowly than cdc13-1 cells even at 25ºC (Figure 2A; top panel). This effect is even more striking upon dissection of a cdc13-1/CDC13 tlc1-tm/TLC1 diploid. We find no difference in the colony size formed by the haploid progeny at 22ºC, regardless of their CDC13 and TLC1 status (Figure 2B). However, cdc13-1 tlc1-tm spores were unable to germinate at 25ºC (Figure 2B), although the cdc13-1 tlc1-tm spores that germinated at 22ºC were able to grow at 25ºC (Figure 2A). These findings suggest that G-quadruplex-mediated capping may be important even at a temperature (25ºC) where the Cdc13-1 mutant protein is only modestly impaired [25].
–
While our findings are consistent with a previously proposed model in which G-quadruplexes protect cdc13-1 telomeres [11], the effect of tlc1-tm on cdc13-1 cells may instead be due to reduced levels of Rap1 at tlc1-tm telomeres [17] rather than a disruption in G-quadruplex formation. However, we do not favor this possibility because telomeres in tlc1-tm cells still retain wild-type telomeric sequence in their centromere-proximal regions, so that telomere-bound Rap1 is only reduced by 40% [17].
DISCUSSION
In this study, we investigated the function of G-quadruplexes at S. cerevisiae telomeres using the tlc1-tm mutant, which causes the addition of mutant telomeric repeats that are defective in forming G-quadruplexes. Our findings suggest that G-quadruplex formation at telomeres is not essential for telomere capping nor cell viability in S. cerevisiae. In addition, our findings are not consistent with a previously proposed model whereby Est1-mediated G-quadruplex formation is required for telomerase activity [10], since tlc1-tm telomeres are efficiently extended by telomerase [17]. While we cannot exclude the possibility that less stable G-quadruplex structures (e.g. two-quartet G-quadruplexes) are able to form at tlc1-tm telomeres, there are other viable tlc1 template mutants that result in telomeric repeats that lack even a double GG motif [18][19]. Nonetheless, our findings are in agreement with a previously proposed model suggesting that telomeric G-quadruplexes serve as capping structures to protect cdc13-1 telomeres [11], and it is also possible that telomeric G-quadruplexes are important for telomere function when S. cerevisiae cells are grown in stress-inducing conditions. Furthermore, we have previously reported several telomeric defects (e.g. disruption of telomere length homeostasis) in tlc1-tm cells [17]. While we believe that most of these defects can be largely attributed to depletion of telomere-bound Rap1, it is formally possible that impairment in the formation of telomeric G-quadruplexes could contribute to some of these defects.
–
The telomere repeats of S. cerevisiae and other Saccharomycotina species are highly divergent and differ from the TTAGGG or TTAGGG-like repeats found in many other eukaryotic species [26][27]. Budding yeast repeats can be quite long, occasionally degenerate, and often non-G/C-rich [28][29]. Many of the budding yeast telomere sequences do not conform to the G≥3NxG≥3NxG≥3NxG≥3 G-quadruplex consensus. Changes in the sequence of the telomeric repeats were accompanied by co-evolution of telomere-binding proteins. In organisms with TTAGGG telomeric repeats, the double-stranded telomeric sequence is typically recognized by proteins homologous to mammalian TRF1 and TRF2, while the single-stranded telomeric sequence is bound by proteins homologous to mammalian POT1. Telomere association of these proteins is highly sequence specific [30][31], so mutating the template region of telomerase RNA leads to a loss of cell viability [32][33][34]. In contrast, the telomeres of Saccharomycotina budding yeast species (with the exception of the Yarrowia clade, one of the basal lineages of Saccharomycotina [35]) are bound by Rap1 and Cdc13. Rap1 and Cdc13 have the possibility to accommodate different target sequences, thereby facilitating the rapid evolution of budding yeast telomeric sequences [29]. A consequence of this rapid evolution may be the loss of a need for telomeric G-quadruplexes. Further studies are needed to determine whether G-quadruplexes are required for proper telomere maintenance in species with TTAGGG telomeric repeats. One recent study has reported that folding of telomeric DNA newly synthesized by human telomerase into G-quadruplexes is important to support telomerase function, which the authors suggest could provide an explanation for the evolutionary conservation of the G-quadruplex-forming potential of telomeric sequence [36]. Addressing this question is especially relevant given that G-quadruplexes have increasingly been proposed as therapeutic targets in oncology [37].
–
If G-quadruplexes are not essential for telomere capping in S. cerevisiae, why does Rap1 have the ability to bind and promote the formation of G-quadruplexes [7][8]? We propose two possible explanations. First, this ability may have been required for telomere capping, but this requirement was lost during the evolution of the Saccharomycotina subdivision. Rudimentary G-quadruplex-based capping in cdc13-1 mutants [11] may be an evolutionary remnant of this requirement, so it would be interesting to test whether suppression of cdc13-1 capping defects by G-quadruplex-stabilizing treatments is dependent on Rap1. Second, the ability of Rap1 to bind and promote the formation of G-quadruplexes may be important for Rap1’s function as a transcriptional regulator [38], rather than for telomere capping. Consistent with this hypothesis, G-quadruplex-forming sequences are strongly enriched at promoters and are thought to influence transcription [39]. These two hypotheses are not mutually exclusive, and it will be interesting to explore their validity in future studies.
MATERIALS AND METHODS
Yeast strains
Standard yeast media and growth conditions were used [40][41]. Yeast strains used in this study are listed in Table 1. Deletion of PIF1 was accomplished by PCR-based gene deletion [42]. Knock-in of the tlc1-tm allele was accomplished by PCR amplification of tlc1-tm from either MCY415 or MCY416, using primers oSMS1 (5′-ACCTGCCTTTGCAGATCCTT-3′) and TLC1-RV (5′-TTATCTTTGGTTCCTTGCCG-3′), followed by transformation of the PCR product into yeast cells using the LiAc-based method [43]. The diploid strain dissected in Figure 2B was generated by knock-in of the tlc1-tm allele into SSY238. The spore colonies were genotyped by replica plating onto YPD + clonNAT plates (to select for the tlc1-tm allele) and YPD plates that were subsequently incubated at 30ºC (to identify cdc13-1 spore colonies, which do not grow at 30ºC).
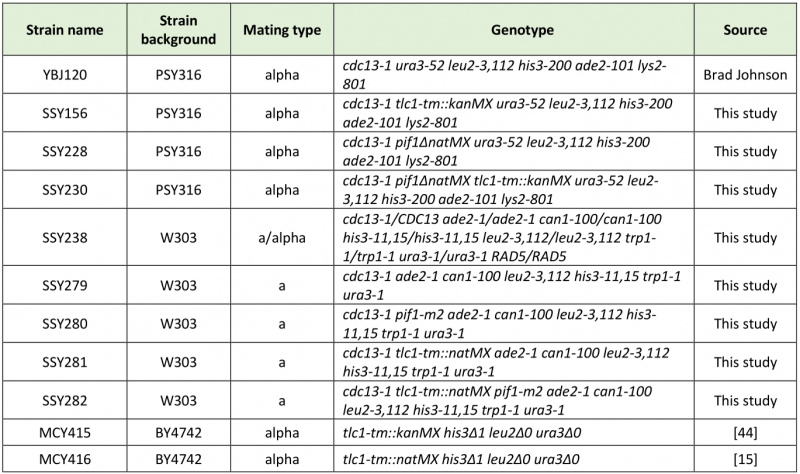 |
TABLE 1. Yeast strains used in this study. |
Spot assays
Cultures for spot assays were grown overnight and diluted to an optical density (OD600) of 0.5, from which four serial 1:10 dilutions were spotted onto YPD plates. Plates were incubated at indicated temperatures for 2 or 3 days.
CD spectroscopy
Oligonucleotides were dissolved in a 10 mM Tris-HCl pH 7.5 and 100 mM KCl solution in a final concentration of 5 μM. The mix was boiled for 5 min at 95ºC and then cooled down overnight. The CD spectra were then measured using a Jasco J-815 spectropolarimeter. Three reads per sample were taken at a wavelength range of 215-350 nm in a quartz cuvette with a 1 cm path length. Data were analyzed using Spekwin32 software.
REFERENCES
- Jain D, Cooper JP (2010). Telomeric strategies: means to an end. Annu Rev Genet 44: 243-269. 10.1146/annurev-genet-102108-134841
- Rhodes D, Lipps HJ (2015). G-quadruplexes and their regulatory roles in biology. Nucleic Acids Res 43(18): 8627-8637. 10.1093/nar/gkv862
- Fang G, Cech TR (1993). The beta subunit of Oxytricha telomere-binding protein promotes G-quartet formation by telomeric DNA. Cell 74(5): 875-885. 10.1016/0092-8674(93)90467-5
- Paeschke K, Simonsson T, Postberg J, Rhodes D, Lipps HJ (2005). Telomere end-binding proteins control the formation of G-quadruplex DNA structures in vivo. Nat Struct Mol Biol 12(10): 847-854. 10.1038/nsmb982
- Paeschke K, Juranek S, Simonsson T, Hempel A, Rhodes D, Lipps HJ (2008). Telomerase recruitment by the telomere end binding protein-beta facilitates G-quadruplex DNA unfolding in ciliates. Nat Struct Mol Biol 15(6): 598-604. 10.1038/nsmb.1422
- Postberg J, Tsytlonok M, Sparvoli D, Rhodes D, Lipps HJ (2012). A telomerase-associated RecQ protein-like helicase resolves telomeric G-quadruplex structures during replication. Gene 497(2): 147-154. 10.1016/j.gene.2012.01.068
- Giraldo R, Rhodes D (1994). The yeast telomere-binding protein RAP1 binds to and promotes the formation of DNA quadruplexes in telomeric DNA. EMBO J 13(10): 2411-2420. 10.1002/j.1460-2075.1994.tb06526.x
- Traczyk A, Liew CW, Gill DJ, Rhodes D (2020). Structural basis of G-quadruplex DNA recognition by the yeast telomeric protein Rap1. Nucleic Acids Res 48(8): 4562-4571. 10.1093/nar/gkaa171
- Jurikova K, Gajarsky M, Hajikazemi M, Nosek J, Prochazkova K, Paeschke K, Trantirek L, Tomaska L (2020). Role of folding kinetics of secondary structures in telomeric G-overhangs in the regulation of telomere maintenance in Saccharomyces cerevisiae. J Biol Chem 295(27): 8958-8971. 10.1074/jbc.RA120.012914
- Zhang ML, Tong XJ, Fu XH, Zhou BO, Wang J, Liao XH, Li QJ, Shen N, Ding J, Zhou JQ (2010). Yeast telomerase subunit Est1p has guanine quadruplex-promoting activity that is required for telomere elongation. Nat Struct Mol Biol 17(2): 202-209. 10.1038/nsmb.1760
- Smith JS, Chen Q, Yatsunyk LA, Nicoludis JM, Garcia MS, Kranaster R, Balasubramanian S, Monchaud D, Teulade-Fichou MP, Abramowitz L, Schultz DC, Johnson FB (2011). Rudimentary G-quadruplex-based telomere capping in Saccharomyces cerevisiae. Nat Struct Mol Biol 18(4): 478-485. 10.1038/nsmb.2033
- Lin JJ, Zakian VA (1996). The SaccharomycesCDC13 protein is a single-strand TG1-3 telomeric DNA-binding protein in vitro that affects telomere behavior in vivo. Proc Natl Acad Sci U S A 93(24): 13760-13765. 10.1073/pnas.93.24.13760
- Nugent CI, Hughes TR, Lue NF, Lundblad V (1996). Cdc13p: a single-strand telomeric DNA-binding protein with a dual role in yeast telomere maintenance. Science 274(5285): 249-252. 10.1126/science.274.5285.249
- Bryan TM (2020). G-Quadruplexes at Telomeres: Friend or Foe? Molecules 25(16). 10.3390/molecules25163686
- Chang M, Arneric M, Lingner J (2007). Telomerase repeat addition processivity is increased at critically short telomeres in a Tel1-dependent manner in Saccharomycescerevisiae. Genes Dev 21(19): 2485-2494. 10.1101/gad.1588807
- Förstemann K, Lingner J (2001). Molecular basis for telomere repeat divergence in budding yeast. Mol Cell Biol 21(21): 7277-7286. 10.1128/MCB.21.21.7277-7286.2001
- Rosas Bringas FR, Stinus S, de Zoeten P, Cohn M, Chang M (2022). Rif2 protects Rap1-depleted telomeres from MRX-mediated degradation in Saccharomyces cerevisiae. eLife 11: e74090. 10.7554/eLife.74090
- Förstemann K, Zaug AJ, Cech TR, Lingner J (2003). Yeast telomerase is specialized for C/A-rich RNA templates. Nucleic Acids Res 31(6): 1646-1655. 10.1093/nar/gkg261
- Lin J, Smith DL, Blackburn EH (2004). Mutant telomere sequences lead to impaired chromosome separation and a unique checkpoint response. Mol Biol Cell 15(4): 1623-1634. 10.1091/mbc.e03-10-0740
- Brázda V, Kolomaznik J, Lýsek J, Bartas M, Fojta M, štastný J, Mergny J-L (2019). G4Hunter web application: a web server for G-quadruplex prediction. Bioinformatics 35(18): 3493-3495. 10.1093/bioinformatics/btz087
- Venczel EA, Sen D (1993). Parallel and antiparallel G-DNA structures from a complex telomeric sequence. Biochemistry 32(24): 6220-6228. 10.1021/bi00075a015
- Paeschke K, Bochman ML, Garcia PD, Cejka P, Friedman KL, Kowalczykowski SC, Zakian VA (2013). Pif1 family helicases suppress genome instability at G-quadruplex motifs. Nature 497(7450): 458-462. 10.1038/nature12149
- Downey M, Houlsworth R, Maringele L, Rollie A, Brehme M, Galicia S, Guillard S, Partington M, Zubko MK, Krogan NJ, Emili A, Greenblatt JF, Harrington L, Lydall D, Durocher D (2006). A genome-wide screen identifies the evolutionarily conserved KEOPS complex as a telomere regulator. Cell 124(6): 1155-1168. 10.1016/j.cell.2005.12.044
- Schulz VP, Zakian VA (1994). The Saccharomyces PIF1 DNA helicase inhibits telomere elongation and de novo telomere formation. Cell 76(1): 145-155. 10.1016/0092-8674(94)90179-1
- Paschini M, Toro TB, Lubin JW, Braunstein-Ballew B, Morris DK, Lundblad V (2012). A naturally thermolabile activity compromises genetic analysis of telomere function in Saccharomyces cerevisiae. Genetics 191(1): 79-93. 10.1534/genetics.111.137869
- Cervenák F, Sepšiová R, Nosek J, Tomáška L (2021). Step-by-Step Evolution of Telomeres: Lessons from Yeasts. Genome Biol Evol 13(2). 10.1093/gbe/evaa268
- Peska V, Fajkus P, Bubeník M, Brázda V, Bohálová N, Dvorácek V, Fajkus J, Garcia S (2021). Extraordinary diversity of telomeres, telomerase RNAs and their template regions in Saccharomycetaceae. Sci Rep 11(1): 12784. 10.1038/s41598-021-92126-x
- Cohn M, McEachern MJ, Blackburn EH (1998). Telomeric sequence diversity within the genus Saccharomyces. Curr Genet 33(2): 83-91. 10.1007/s002940050312
- Steinberg-Neifach O, Lue NF (2015). Telomere DNA recognition in Saccharomycotina yeast: potential lessons for the co-evolution of ssDNA and dsDNA-binding proteins and their target sites. Front Genet 6: 162. 10.3389/fgene.2015.00162
- Broccoli D, Smogorzewska A, Chong L, de Lange T (1997). Human telomeres contain two distinct Myb-related proteins, TRF1 and TRF2. Nat Genet 17(2): 231-235. 10.1038/ng1097-231
- Loayza D, Parsons H, Donigian J, Hoke K, de Lange T (2004). DNA binding features of human POT1: a nonamer 5’-TAGGGTTAG-3’ minimal binding site, sequence specificity, and internal binding to multimeric sites. J Biol Chem 279(13): 13241-13248. 10.1074/jbc.M312309200
- Marušic L, Anton M, Tidy A, Wang P, Villeponteau B, Bacchetti S (1997). Reprogramming of telomerase by expression of mutant telomerase RNA template in human cells leads to altered telomeres that correlate with reduced cell viability. Mol Cell Biol 17(11): 6394-6401. 10.1128/mcb.17.11.6394
- Guiducci C, Cerone MA, Bacchetti S (2001). Expression of mutant telomerase in immortal telomerase-negative human cells results in cell cycle deregulation, nuclear and chromosomal abnormalities and rapid loss of viability. Oncogene 20(6): 714-725. 10.1038/sj.onc.1204145
- Kim MM, Rivera MA, Botchkina IL, Shalaby R, Thor AD, Blackburn EH (2001). A low threshold level of expression of mutant-template telomerase RNA inhibits human tumor cell proliferation. Proc Natl Acad Sci U S A 98(14): 7982-7987. 10.1073/pnas.131211098
- Cervenák F, Juríková K, Devillers H, Kaffe B, Khatib A, Bonnell E, Sopkovicová M, Wellinger RJ, Nosek J, Tzfati Y, Neuvéglise C, Tomáška L (2019). Identification of telomerase RNAs in species of the Yarrowia clade provides insights into the co-evolution of telomerase, telomeric repeats and telomere-binding proteins. Sci Rep 9(1): 13365. 10.1038/s41598-019-49628-6
- Jansson LI, Hentschel J, Parks JW, Chang TR, Lu C, Baral R, Bagshaw CR, Stone MD (2019). Telomere DNA G-quadruplex folding within actively extending human telomerase. Proc Natl Acad Sci U S A 116(19): 9350-9359. 10.1073/pnas.1814777116
- Balasubramanian S, Hurley LH, Neidle S (2011). Targeting G-quadruplexes in gene promoters: a novel anticancer strategy? Nat Rev Drug Discov 10(4): 261-275. 10.1038/nrd3428
- Azad GK, Tomar RS (2016). The multifunctional transcription factor Rap1: a regulator of yeast physiology. Front Biosci (Landmark Ed) 21: 918-930. 10.2741/4429
- Varshney D, Spiegel J, Zyner K, Tannahill D, Balasubramanian S (2020). The regulation and functions of DNA and RNA G-quadruplexes. Nat Rev Mol Cell Biol 21(8): 459-474. 10.1038/s41580-020-0236-x
- Sherman F (2002). Getting started with yeast. Methods Enzymol 350: 3-41. 10.1016/s0076-6879(02)50954-x
- Lundblad V, Struhl K (2010). Yeast. Curr. Protoc. Mol. Biol. 92(1). 10.1002/0471142727.mb1300s92
- Goldstein AL, McCusker JH (1999). Three new dominant drug resistance cassettes for gene disruption in Saccharomyces cerevisiae. Yeast 15(14): 1541-1553. 10.1002/(SICI)1097-0061(199910)15:14<1541::AID-YEA476>3.0.CO;2-K
- Schiestl RH, Gietz RD (1989). High efficiency transformation of intact yeast cells using single stranded nucleic acids as a carrier. Curr Genet 16(5-6): 339-346. 10.1007/BF00340712
- Strecker J, Stinus S, Pliego Caballero M, Szilard RK, Chang M, Durocher D (2017). A sharp Pif1-dependent threshold separates DNA double-strand breaks from critically short telomeres. eLife 6: e23783. 10.7554/eLife.23783
–
ACKNOWLEDGMENTS
We thank M. Cohn, K. Paeschke, and M. Hajikazemi for critical reading of the manuscript, and H.G. Kazemier for technical assistance. F.R.R.B. was supported by a CONACYT scholarship.
COPYRIGHT
© 2022

Investigating the role of G-quadruplexes at Saccharomyces cerevisiae telomeres by Stinus et al. is licensed under a Creative Commons Attribution 4.0 International License.