Back to article: The last two transmembrane helices in the APC-type FurE transporter act as an intramolecular chaperone essential for concentrative ER-exit
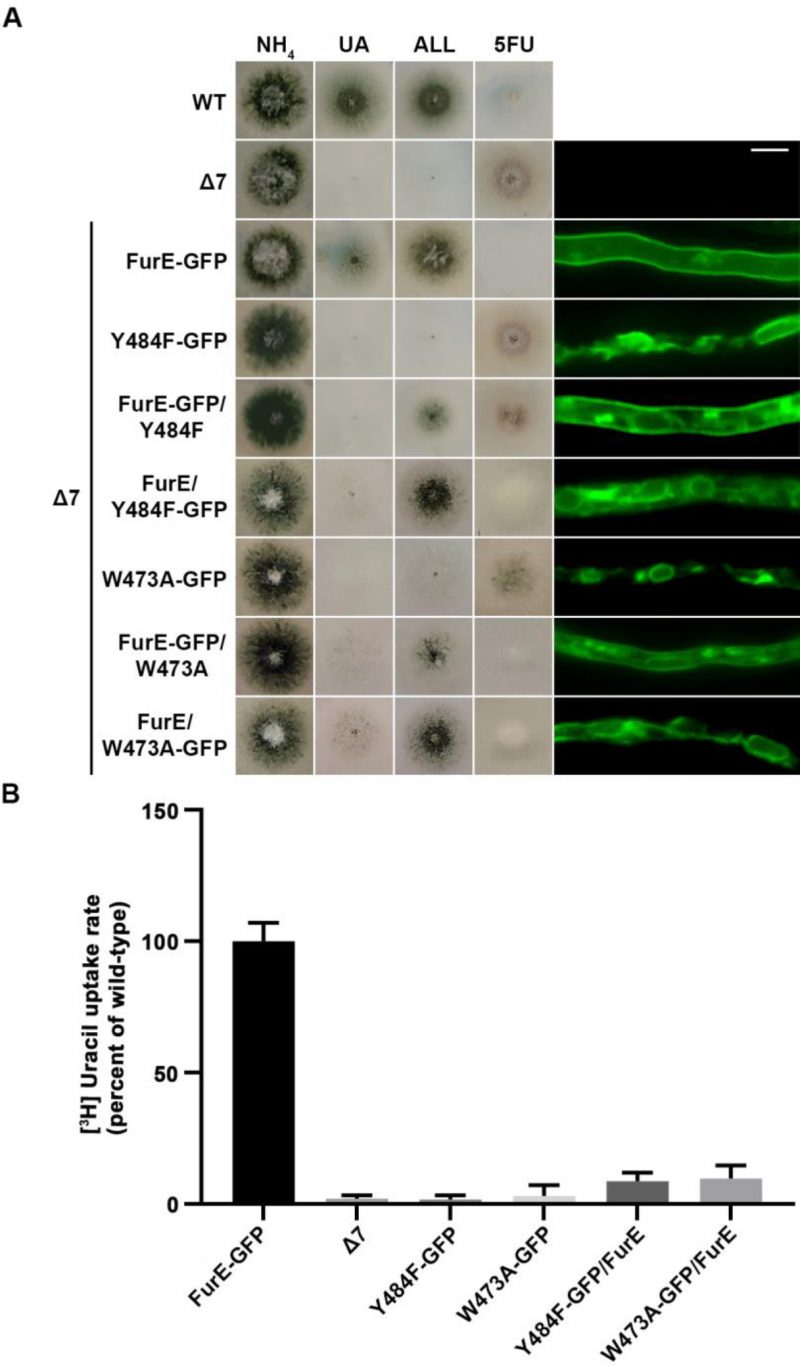
FIGURE 5: ER-retained mutants affect wild-type FurE localization, resulting in reduced transport activity. (A) Growth tests and epifluorescence microscopy of controls and strains co-expressing wild-type FurE with ER-retained FurE mutant versions. GFP-tagging is either on the wt FurE or in the ER-retained mutant. Notice the appearance of fluorescent signal associated with ER membranes (mostly as perinuclear rings) in the case were wt FurE-GFP is co-expressed with untagged Y484F or W473A. Scale bar corresponds to 5μM. (B) Radiolabeled uracil uptake rates, expressed as % of wt FurE rate, of strains expressing FurE mutant versions. For details see Materials and Methods.