Back to article: Sulfur transfer and activation by ubiquitin-like modifier system Uba4•Urm1 link protein urmylation and tRNA thiolation in yeast
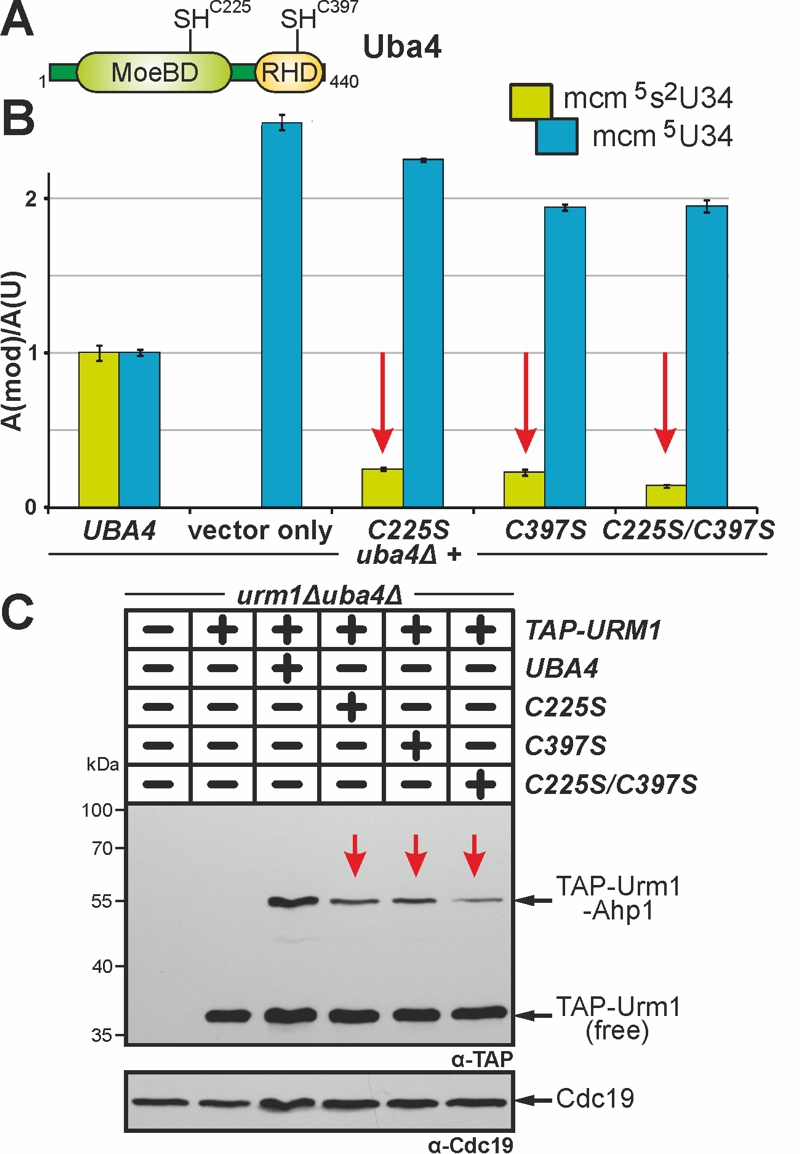
FIGURE 4: Cys225 and Cys397 are critical but not essential for Uba4 to activate Urm1 functions in tRNA thiolation and urmylation.
(A) Uba4 scheme depicting MoeBD and RHD regions and the positions of Cys225 and Cys397.
(B) tRNA (thio)modification analysis via LC-MS/MS (see Fig. 3) reveals Cys225 and/or Cys397 substitution mutants significantly interfere (red arrows) with formation of the mcm5s2U34 modification.
(C) Ahp1 urmylation is strongly (red arrows) affected by the C225S, C397S and C225S/C397S mutants. Urmylation assays (see Fig. 2) involved anti-TAP-based EMSA to detect non-conjugated (free) TAP-Urm1 and TAP-Urm1•Ahp1 conjugates. Protein loading was controlled with anti-Cdc19 antibodies.