
Regulation of extracellular vesicles for protein secretion in Aspergillus nidulans
This study reveals that Aspergillus nidulans boosts extracellular vesicle production when ER-trafficked enzymes are induced, uncovering how fungi remodel their secretome through vesicle-mediated secretion to adapt to changing environments and biofilm formation.

Transcriptomic response to different heme sources in Trypanosoma cruzi epimastigotes
This study uncovers how the Chagas disease parasite adapts to changes in heme, an essential molecule for its survival, providing transcriptional clues to heme metabolism and identifying a previously unreported heme-binding protein in T. cruzi.

Luminal acetylation of microtubules is not essential for Plasmodium berghei and Toxoplasma gondii survival
Acetylation of α-tubulin at lysine 40 is not essential for cytoskeletal stability in Plasmodium berghei or Toxoplasma gondii, suggesting redundancy and plasticity in microtubule regulation in these parasites.

The dual-site agonist for human M2 muscarinic receptors Iper-8-naphtalimide induces mitochondrial dysfunction in Saccharomyces cerevisiae
S. cerevisiae is a model to study human GPCRs. N-8-Iper, active against glioblastoma via M2 receptor, causes mitochondrial damage in yeast by binding Ste2, highlighting evolutionary conservation of GPCRs.

Integrative Omics reveals changes in the cellular landscape of peroxisome-deficient pex3 yeast cells
To uncover the consequences of peroxisome deficiency, we compared Saccharomyces cerevisiae wild-type with pex3 cells, which lack peroxisomes, employing quantitative proteomics and transcriptomics technologies.

Regulation of extracellular vesicles for protein secretion in Aspergillus nidulans
Rebekkah E. Pope1, Patrick Ballmann2, Lisa Whitworth3 and Rolf A. Prade1,*
This study reveals that Aspergillus nidulans boosts extracellular vesicle production when ER-trafficked enzymes are induced, uncovering how fungi remodel their secretome through vesicle-mediated secretion to adapt to changing environments and biofilm formation.

Transcriptomic response to different heme sources in Trypanosoma cruzi epimastigotes
Evelyn Tevere1,a, María G. Mediavilla1,a, Cecilia B. Di Capua1, Marcelo L. Merli1, Carlos Robello2,3, Luisa Berná2,4 and Julia A. Cricco
This study uncovers how the Chagas disease parasite adapts to changes in heme, an essential molecule for its survival, providing transcriptional clues to heme metabolism and identifying a previously unreported heme-binding protein in T. cruzi.
Sir2 regulates selective autophagy in stationary-phase yeast cells
Ji-In Ryua, Juhye Junga, and Jeong-Yoon Kim
This study establishes Sir2 as a previously unrecognized regulator of selective autophagy during the stationary phase and highlight how cells dynamically control organelle degradation.
Global translational impacts of the loss of the tRNA modification t6A in yeast
Patrick C. Thiaville1,2,3,4, Rachel Legendre4, Diego Rojas-Benítez5, Agnès Baudin-Baillieu4, Isabelle Hatin4, Guilhem Chalancon6, Alvaro Glavic5, Olivier Namy4, Valérie de Crécy-Lagard1,3
The universal tRNA modification t6A is found at position 37 of nearly all tRNAs decoding ANN codons. Analysis of codon occupancy rates suggests that one of the major roles of t6A is to homogenize the process of elongation by slowing the elongation rate at codons decoded by high abundance tRNAs and I34:C3 pairs while increasing the elongation rate of rare tRNAs and G34:U3 pairs. This work reveals that the consequences of t6A absence are complex and multilayered and has set the stage to elucidate the molecular basis of the observed phenotypes.
Ergosterone-coupled Triazol molecules trigger mitochondrial dysfunction, oxidative stress, and acidocalcisomal Ca2+ release in Leishmania mexicana promastigotes
Figarella K1, Marsiccobetre S1, Arocha I1, Colina W2, Hasegawa M2,†, Rodriguez M2, Rodriguez-Acosta A3, Duszenko M4, Benaim G5, Uzcategui NL3
The protozoan parasite Leishmania causes a variety of sicknesses with different clinical manifestations known as leishmaniasis. Investigations looking for new targets or new active molecules focus mainly on the disruption of parasite specific pathways. In this sense, ergosterol biosynthesis is one of the most attractive because it does not occur in mammals. Our results indicate that ergosterone-triazol coupled molecules induce a regulated cell death process in the parasite and may represent starting point molecules in the search of new chemotherapeutic agents to combat leishmaniasis.
INO1 transcriptional memory leads to DNA zip code-dependent interchromosomal clustering
Donna Garvey Brickner, Robert Coukos and Jason H. Brickner
Many genes localize at the nuclear periphery through physical interaction with the nuclear pore complex (NPC). We have found that the yeast INO1 gene is targeted to the NPC both upon activation and for several generations after repression, a phenomenon called epigenetic transcriptional memory. Targeting of INO1 to the NPC requires distinct cis-acting promoter DNA zip codes under activating conditions and under memory conditions. When at the nuclear periphery, active INO1 clusters with itself and with other genes that share the GRS I zip code. Here, we show that during memory, the two alleles of INO1 cluster in diploids and endogenous INO1 clusters with an ectopic INO1 in haploids. After repression, INO1 does not cluster with GRS I – containing genes. Furthermore, clustering during memory requires Nup100 and two sets of DNA zip codes…
A central role for TOR signalling in a yeast model for juvenile CLN3 disease
Michael E. Bond1, Rachel Brown1, Charalampos Rallis3,4, Jürg Bähler3,4 and Sara E. Mole1,2,3
Yeasts provide an excellent genetically tractable eukaryotic system for investigating the function of genes in their biological context, and are especially relevant for those conserved genes that cause disease. Bond et al. study the role of btn1, the orthologue of a human gene that underlies an early onset neurodegenerative disease (juvenile CLN3 disease, neuronal ceroid lipofuscinosis (NCLs) or Batten disease) in the fission yeast Schizosaccharomyces pombe.
Oxygen availability strongly affects chronological lifespan and thermotolerance in batch cultures of Saccharomyces cerevisiae
Markus M.M. Bisschops1,3,#, Tim Vos1,#, Rubén Martínez-Moreno2,4, Pilar de la Torre Cortés1, Jack T. Pronk1, Pascale Daran-Lapujade1
Stationary-phase (SP) batch cultures of Saccharomyces cerevisiae, in which growth has been arrested by carbon-source depletion, are widely applied to study chronological lifespan, quiescence and SP-associated robustness. Based on this type of experiments, typically performed under aerobic conditions, several roles of oxygen in aging have been proposed. However, SP in anaerobic yeast cultures has not been investigated in detail. Here, we use the unique capability of S. cerevisiae to grow in the complete absence of oxygen to directly compare SP in aerobic and anaerobic bioreactor cultures. This comparison revealed strong positive effects of oxygen availability on adenylate energy charge, longevity and thermotolerance during SP. A low thermotolerance of…
DNA damage checkpoint adaptation genes are required for division of cells harbouring eroded telomeres
Sofiane Y. Mersaoui, Serge Gravel, Victor Karpov, and Raymund J. Wellinger
In budding yeast, telomerase and the Cdc13p protein are two key players acting to ensure telomere stability. This article shows that while the capping process can be flexible, it takes a very specific genetic setup to allow a change from canonical capping to alternative capping.
The MAPKKKs Ste11 and Bck1 jointly transduce the high oxidative stress signal through the cell wall integrity MAP kinase pathway
Chunyan Jin#, Stephen K. Kim, Stephen D. Willis and Katrina F. Cooper
Oxidative stress stimulates the Rho1 GTPase, which in turn induces the cell wall integrity (CWI) MAP kinase cascade. CWI activation promotes stress-responsive gene expression through activation of transcription factors (Rlm1, SBF) and nuclear release and subsequent destruction of the repressor cyclin C. This study reports that, in response to high hydrogen peroxide exposure, or in the presence of constitutively active Rho1, cyclin C still translocates to the cytoplasm and is degraded in cells lacking Bck1, the MAPKKK of the CWI pathway.
Formyl-methionine as a degradation signal at the N-termini of bacterial proteins
Konstantin I. Piatkov1,3,#, Tri T. M. Vu1,#, Cheol-Sang Hwang2 and Alexander Varshavsky1
Varshavsky and colleagues solve a long-standing mystery in proteolysis! In bacteria, all nascent proteins bear the pretranslationally formed N-terminal formyl-methionine (fMet) residue. The fMet residue is cotranslationally deformylated by a ribosome-associated deformylase. The formylation of N-terminal Met in bacterial proteins is not strictly essential for either translation or cell viability. Moreover, protein synthesis by the cytosolic ribosomes of eukaryotes does not involve the formylation of N-terminal Met. What, then, is the main biological function of this metabolically costly, transient, and not strictly essential modification of N‑terminal Met, and why has Met formylation not been eliminated during bacterial evolution? One possibility is that the similarity of the formyl and acetyl groups, their identical locations in…
Modeling non-hereditary mechanisms of Alzheimer disease during apoptosis in yeast
Ralf J. Braun1,#, Cornelia Sommer2,3,#, Christine Leibiger1,#, Romina J.G. Gentier4,#, Verónica I. Dumit5, Katrin Paduch1, Tobias Eisenberg2, Lukas Habernig2, Gert Trausinger6, Christoph Magnes6, Thomas Pieber6,7, Frank Sinner6,7, Jörn Dengjel5, Fred W. van Leeuwen4, Guido Kroemer8-11, and Frank Madeo2,3
Impaired protein degradation and mitochondrial dysfunction are believed to contribute to neurodegenerative disorders, including Alzheimer disease (AD). This microreview comments on the article “Accumulation of Basic Amino Acids at Mitochondria Dictates the Cytotoxicity of Aberrant Ubiquitin” by Braun et al. (2015), Cell Rep.
Translate to divide: сontrol of the cell cycle by protein synthesis
Michael Polymenis1 and Rodolfo Aramayo2
Protein synthesis underpins much of cell growth and, consequently, cell multiplication. Understanding how proliferating cells commit and progress into the cell cycle requires knowing not only which proteins need to be synthesized, but also what determines their rate of synthesis during cell division. Experiments with proliferating populations of microbial strains, animal or plant cell lines, have rigorous expectations. Under the same culture conditions, cells ought to have the same properties and composition in every single experiment. The basic “metrics” of proliferating cells remain constant, even after many rounds of cell division. These metrics include cellular mass and volume, and macromolecular composition. The constancy of such parameters reflects the fundamental ability of cells to coordinate their growth with their division. Balancing cell growth with cell division determines the overall rates of cell proliferation…
New roles for autophagy and spermidine in T cells
D. J. Puleston and A. K. Simon
This microreview discusses the article “Autophagy is a critical regulator of memory CD8+ T cell formation” by Puleston et al. (2014), eLife.
Characterization of the Maf family of polymorphic toxins in pathogenic Neisseria species
Anne Jamet1,2,3,4,5, Xavier Nassif2,3,4,5
In addition to harmless commensal species, Neisseria genus encompasses two pathogenic species, N. meningitidis (the meningococcus) and N. gonorrhoeae (the gonococcus), which are responsible for meningitis and genital tract infections, respectively. This microreview comments on the article “A new family of secreted toxins in pathogenic Neisseria species” by Jamet et al. (2015), PLoS Pathog.
Live fast, die soon: cell cycle progression and lifespan in yeast cells
Javier Jiménez, Samuel Bru, Mariana PC Ribeiro and Josep Clotet
Our understanding of lifespan has benefited enormously from the study of a simple model, the yeast Saccharomyces cerevisiae. Although a unicellular organism, yeasts undergo many of the processes directly related with aging that to some extent are conserved in mammalian cells. Nutrient-limiting conditions have been involved in lifespan extension, especially in the case of caloric restriction, which also has a direct impact on cell cycle progression. In fact, other environmental stresses (osmotic, oxidative) that interfere with normal cell cycle progression also influence the lifespan of cells, indicating a relationship between lifespan and cell cycle control. In the present review we compile and discuss new findings related to how cell cycle progression is regulated by other nutrients. We centred this review on the analysis of phosphate, also give some attention to nitrogen, and the impact of these nutrients on lifespan…
Yeast as a tool for studying proteins of the Bcl-2 family
Peter Polčic, Petra Jaká and Marek Mentel
This review focuses on using yeast expressing mammalian proteins of the Bcl-2 family as a tool to investigate mechanisms, by which these proteins permeabilize mitochondrial membranes, mechanisms, by which pro- and antiapoptotic members of this family interact, and involvement of other cellular components in the regulation of programmed cell death by Bcl-2 family proteins.
Mitochondrial type II NAD(P)H dehydrogenases in fungal cell death
Pedro Gonçalves1,2,4, Arnaldo Videira1,2,3
During aerobic respiration, cells produce energy through oxidative phosphorylation, which includes a specialized group of multi-subunit complexes in the inner mitochondrial membrane known as the electron transport chain. However, this canonical pathway is branched into single polypeptide alternative routes in some fungi, plants, protists and bacteria. They confer metabolic plasticity, allowing cells to adapt to different environmental conditions and stresses…
EzrA: a spectrin-like scaffold in the bacterial cell division machinery
Robert M Cleverley, Richard J Lewis
Much progress has been made in identifying the components of the divisome, the assembly of proteins that undertakes the vital process of cell division in bacteria. However, how the highly interdependent processes on either side of the membrane are coordinated during division is a major unresolved question. This comment discusses the article “Structure and function of a spectrin-like regulator of bacterial cytokinesis” by Cleverley et al. (2014), Nat Commun.
Microbial hara-kiri: Exploiting lysosomal cell death in malaria parasites
Jun-Hong Ch’ng1,2, Johan Ursing2 and Kevin Shyong-Wei Tan1
The antimalarial drug chloroquine (CQ) has been sidelined in the fight against falciparum malaria due to wide-spread CQ resistance. This comment discusses the article “Validation of a chloroquine-induced cell death mechanism for clinical use against malaria” by Ch’ng et al. (2014), Cell Death Dis.
The emerging role of complex modifications of tRNALysUUU in signaling pathways
Patrick C. Thiaville1,2,3,4 and Valérie de Crécy-Lagard2,4
This comment discusses the article “Loss of wobble uridine modification in tRNA anticodons interferes with TOR pathway signaling” by Scheidt et al (Microbial Cell, 2014).
Only functional localization is faithful localization
Roland Lill1,2,3
This article comments on work published by Peleh et al. (Microbial Cell 2014), which analyzes the localization of Dre2 in Saccharomyces cerevisiae.
One cell, one love: a journal for microbial research
Didac Carmona-Gutierrez1, Guido Kroemer2-6 and Frank Madeo1
In this inaugural article of Microbial Cell, we highlight the importance of microbial research in general and the journal’s intention to serve as a publishing forum that supports and enfolds the scientific diversity in this area as it provides a unique, high-quality and universally accessible source of information and inspiration.
What’s the role of autophagy in trypanosomes?
Katherine Figarella1 and Néstor L. Uzcátegui1,2
This article comments on Proto et al. (Microbial Cell, 2014), who report first insights into the molecular mechanism of autophagy in African trypanosomes by generating reporter bloodstream form cell lines.
Microbial Cell
is an open-access, peer-reviewed journal that publishes exceptionally relevant research works that implement the use of unicellular organisms (and multicellular microorganisms) to understand cellular responses to internal and external stimuli and/or human diseases.
you can trust
Can’t find what you’re looking for?
You can browse all our issues and published articles here.
FAQs
Peer-reviewed, open-access research using unicellular organisms (and multicellular microorganisms) to understand cellular responses and human disease.
The journal (founded in 2014) is led by its Editors-in-Chief Frank Madeo, Didac Carmona-Gutierrez, and Guido Kroemer
Microbial Cell has been publishing original scientific literature since 2014, and from the very beginning has been managed by active scientists through an independent Publishing House (Shared science Publishers). The journal was conceived as a platform to acknowledge the importance of unicellular organisms, both as model systems as well as in the biological context of human health and disease.
Ever since, Microbial Cell has very positively developed and strongly grown into a respected journal in the unicellular research community and even beyond. This scientific impact is reflected in the yearly number of citations obtained by articles published in Microbial Cell, as recorded by the Web of Science (Clarivate, formerly Thomson/Reuters):
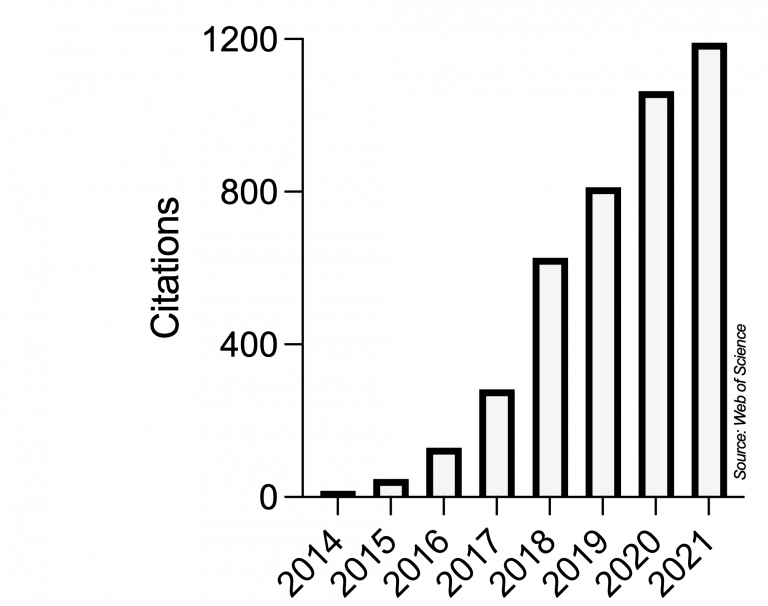
The scientific impact of Microbial Cell is also mirrored in a series of milestones:
2015: Microbial Cell is included in the Emerging Sources Citation Index (ESCI), a selection of developing journals drafted by Clarivate Analytics based on the candidate’s publishing standards, quality, editorial content, and citation data. Note: As an ESCI-selected journal, Microbial Cell is currently being evaluated in a rigorous and long process to determine an inclusion in the Science Citation Index Expanded (SCIE), which allows the official calculation of Clarivate Analytics’ impact factor.
2016: Microbial Cell is awarded the so-called DOAJ Seal by the selective Directory of Open Access Journals (DOAJ). The DOAJ Seal is an exclusive mark of certification for open access journals granted by DOAJ to journals that adhere to outstanding best practice and achieve an extra high and clear commitment to open access and high publishing standards.
2017: Microbial Cell is included in Pubmed Central (PMC), allowing the archiving of all the journal’s articles in PMC and PubMed.
2019: Microbial Cell is indexed in the prestigious abstract and citation database Scopus after a thorough selection process. This also means that Microbial Cell obtains, for the first time, an official Scopus CiteScore as well as an official journal ranking in the Scimago Journal and Country Ranking.
2022: Microbial Cell’s CiteScore reaches a value of 7.2 for the year 2021, positioning Microbial Cell among the top microbiology journals (previously available CiteScores: 2019: 5.4; 2020: 5.1).
2022: Microbial Cell is indexed in the highly selective Science Citation Index Expanded™, which covers approx. 9,500 of the world’s most impactful journals across 178 scientific disciplines. In their journal selection and curation process, Clarivate´s editors apply 24 ‘quality’ criteria and four ‘impact’ criteria to select the most influential journals in their respective fields. This selection is also a pre-requisite for inclusion in the JCR, which features the impact factor.
2022: Microbial Cell is listed in the Journal Citation Reports™ (JCR), and obtains its first official Journal Impact Factor™ (JIF) for the year 2021: 5.316.Check Article Types and Manuscript Preparation guidelines. Submit online via Scholastica.

Metabolic pathways further increase the complexity of cell size control in budding yeast
Jorrit M. Enserink
This article comments on work published by Soma et al. (Microbial Cell, 2014), which teased apart the effect of metabolism and growth rate on setting of critical cell size in Saccharomyces cerevisiae.