
Regulation of extracellular vesicles for protein secretion in Aspergillus nidulans
This study reveals that Aspergillus nidulans boosts extracellular vesicle production when ER-trafficked enzymes are induced, uncovering how fungi remodel their secretome through vesicle-mediated secretion to adapt to changing environments and biofilm formation.

Transcriptomic response to different heme sources in Trypanosoma cruzi epimastigotes
This study uncovers how the Chagas disease parasite adapts to changes in heme, an essential molecule for its survival, providing transcriptional clues to heme metabolism and identifying a previously unreported heme-binding protein in T. cruzi.

Luminal acetylation of microtubules is not essential for Plasmodium berghei and Toxoplasma gondii survival
Acetylation of α-tubulin at lysine 40 is not essential for cytoskeletal stability in Plasmodium berghei or Toxoplasma gondii, suggesting redundancy and plasticity in microtubule regulation in these parasites.

The dual-site agonist for human M2 muscarinic receptors Iper-8-naphtalimide induces mitochondrial dysfunction in Saccharomyces cerevisiae
S. cerevisiae is a model to study human GPCRs. N-8-Iper, active against glioblastoma via M2 receptor, causes mitochondrial damage in yeast by binding Ste2, highlighting evolutionary conservation of GPCRs.

Integrative Omics reveals changes in the cellular landscape of peroxisome-deficient pex3 yeast cells
To uncover the consequences of peroxisome deficiency, we compared Saccharomyces cerevisiae wild-type with pex3 cells, which lack peroxisomes, employing quantitative proteomics and transcriptomics technologies.

Regulation of extracellular vesicles for protein secretion in Aspergillus nidulans
Rebekkah E. Pope1, Patrick Ballmann2, Lisa Whitworth3 and Rolf A. Prade1,*
This study reveals that Aspergillus nidulans boosts extracellular vesicle production when ER-trafficked enzymes are induced, uncovering how fungi remodel their secretome through vesicle-mediated secretion to adapt to changing environments and biofilm formation.

Transcriptomic response to different heme sources in Trypanosoma cruzi epimastigotes
Evelyn Tevere1,a, María G. Mediavilla1,a, Cecilia B. Di Capua1, Marcelo L. Merli1, Carlos Robello2,3, Luisa Berná2,4 and Julia A. Cricco
This study uncovers how the Chagas disease parasite adapts to changes in heme, an essential molecule for its survival, providing transcriptional clues to heme metabolism and identifying a previously unreported heme-binding protein in T. cruzi.
Sir2 regulates selective autophagy in stationary-phase yeast cells
Ji-In Ryua, Juhye Junga, and Jeong-Yoon Kim
This study establishes Sir2 as a previously unrecognized regulator of selective autophagy during the stationary phase and highlight how cells dynamically control organelle degradation.
Global translational impacts of the loss of the tRNA modification t6A in yeast
Patrick C. Thiaville1,2,3,4, Rachel Legendre4, Diego Rojas-Benítez5, Agnès Baudin-Baillieu4, Isabelle Hatin4, Guilhem Chalancon6, Alvaro Glavic5, Olivier Namy4, Valérie de Crécy-Lagard1,3
The universal tRNA modification t6A is found at position 37 of nearly all tRNAs decoding ANN codons. Analysis of codon occupancy rates suggests that one of the major roles of t6A is to homogenize the process of elongation by slowing the elongation rate at codons decoded by high abundance tRNAs and I34:C3 pairs while increasing the elongation rate of rare tRNAs and G34:U3 pairs. This work reveals that the consequences of t6A absence are complex and multilayered and has set the stage to elucidate the molecular basis of the observed phenotypes.
Ergosterone-coupled Triazol molecules trigger mitochondrial dysfunction, oxidative stress, and acidocalcisomal Ca2+ release in Leishmania mexicana promastigotes
Figarella K1, Marsiccobetre S1, Arocha I1, Colina W2, Hasegawa M2,†, Rodriguez M2, Rodriguez-Acosta A3, Duszenko M4, Benaim G5, Uzcategui NL3
The protozoan parasite Leishmania causes a variety of sicknesses with different clinical manifestations known as leishmaniasis. Investigations looking for new targets or new active molecules focus mainly on the disruption of parasite specific pathways. In this sense, ergosterol biosynthesis is one of the most attractive because it does not occur in mammals. Our results indicate that ergosterone-triazol coupled molecules induce a regulated cell death process in the parasite and may represent starting point molecules in the search of new chemotherapeutic agents to combat leishmaniasis.
INO1 transcriptional memory leads to DNA zip code-dependent interchromosomal clustering
Donna Garvey Brickner, Robert Coukos and Jason H. Brickner
Many genes localize at the nuclear periphery through physical interaction with the nuclear pore complex (NPC). We have found that the yeast INO1 gene is targeted to the NPC both upon activation and for several generations after repression, a phenomenon called epigenetic transcriptional memory. Targeting of INO1 to the NPC requires distinct cis-acting promoter DNA zip codes under activating conditions and under memory conditions. When at the nuclear periphery, active INO1 clusters with itself and with other genes that share the GRS I zip code. Here, we show that during memory, the two alleles of INO1 cluster in diploids and endogenous INO1 clusters with an ectopic INO1 in haploids. After repression, INO1 does not cluster with GRS I – containing genes. Furthermore, clustering during memory requires Nup100 and two sets of DNA zip codes…
A central role for TOR signalling in a yeast model for juvenile CLN3 disease
Michael E. Bond1, Rachel Brown1, Charalampos Rallis3,4, Jürg Bähler3,4 and Sara E. Mole1,2,3
Yeasts provide an excellent genetically tractable eukaryotic system for investigating the function of genes in their biological context, and are especially relevant for those conserved genes that cause disease. Bond et al. study the role of btn1, the orthologue of a human gene that underlies an early onset neurodegenerative disease (juvenile CLN3 disease, neuronal ceroid lipofuscinosis (NCLs) or Batten disease) in the fission yeast Schizosaccharomyces pombe.
Oxygen availability strongly affects chronological lifespan and thermotolerance in batch cultures of Saccharomyces cerevisiae
Markus M.M. Bisschops1,3,#, Tim Vos1,#, Rubén Martínez-Moreno2,4, Pilar de la Torre Cortés1, Jack T. Pronk1, Pascale Daran-Lapujade1
Stationary-phase (SP) batch cultures of Saccharomyces cerevisiae, in which growth has been arrested by carbon-source depletion, are widely applied to study chronological lifespan, quiescence and SP-associated robustness. Based on this type of experiments, typically performed under aerobic conditions, several roles of oxygen in aging have been proposed. However, SP in anaerobic yeast cultures has not been investigated in detail. Here, we use the unique capability of S. cerevisiae to grow in the complete absence of oxygen to directly compare SP in aerobic and anaerobic bioreactor cultures. This comparison revealed strong positive effects of oxygen availability on adenylate energy charge, longevity and thermotolerance during SP. A low thermotolerance of…
DNA damage checkpoint adaptation genes are required for division of cells harbouring eroded telomeres
Sofiane Y. Mersaoui, Serge Gravel, Victor Karpov, and Raymund J. Wellinger
In budding yeast, telomerase and the Cdc13p protein are two key players acting to ensure telomere stability. This article shows that while the capping process can be flexible, it takes a very specific genetic setup to allow a change from canonical capping to alternative capping.
The MAPKKKs Ste11 and Bck1 jointly transduce the high oxidative stress signal through the cell wall integrity MAP kinase pathway
Chunyan Jin#, Stephen K. Kim, Stephen D. Willis and Katrina F. Cooper
Oxidative stress stimulates the Rho1 GTPase, which in turn induces the cell wall integrity (CWI) MAP kinase cascade. CWI activation promotes stress-responsive gene expression through activation of transcription factors (Rlm1, SBF) and nuclear release and subsequent destruction of the repressor cyclin C. This study reports that, in response to high hydrogen peroxide exposure, or in the presence of constitutively active Rho1, cyclin C still translocates to the cytoplasm and is degraded in cells lacking Bck1, the MAPKKK of the CWI pathway.
Formyl-methionine as a degradation signal at the N-termini of bacterial proteins
Konstantin I. Piatkov1,3,#, Tri T. M. Vu1,#, Cheol-Sang Hwang2 and Alexander Varshavsky1
Varshavsky and colleagues solve a long-standing mystery in proteolysis! In bacteria, all nascent proteins bear the pretranslationally formed N-terminal formyl-methionine (fMet) residue. The fMet residue is cotranslationally deformylated by a ribosome-associated deformylase. The formylation of N-terminal Met in bacterial proteins is not strictly essential for either translation or cell viability. Moreover, protein synthesis by the cytosolic ribosomes of eukaryotes does not involve the formylation of N-terminal Met. What, then, is the main biological function of this metabolically costly, transient, and not strictly essential modification of N‑terminal Met, and why has Met formylation not been eliminated during bacterial evolution? One possibility is that the similarity of the formyl and acetyl groups, their identical locations in…
A roadmap for designing narrow-spectrum antibiotics targeting bacterial pathogens
Xinyun Cao1,*, Robert Landick1,2, Elizabeth A. Campbell3
This comment discusses the article “Basis of narrow-spectrum activity of fidaxomicin on Clostridioides difficile” by Cao et al. (2022, Nature).
Breaking the clip for cargo unloading from motor proteins: mechanism and significance
Keisuke Obara1, and Takumi Kamura1
The mitochondrion is an essential organelle involved in ATP generation, lipid metabolism, regulation of calcium ions, etc. Therefore, it should be inherited properly by newly generated cells. In the budding yeast Saccharomyces cerevisiae, mitochondria are passed on to daughter cells by the motor protein, Myo2, on the actin cable. The mitochondria and Myo2 are connected via the adaptor protein Mmr1. After reaching daughter cells, mitochondria are released from the actin-myosin machinery and move dynamically. In our recent paper (Obara K et al. (2022), Nat Commun, doi:10.1038/s41467-022-29704-8), we demonstrated that the regulated proteolysis of Mmr1 is required for the unloading of mitochondria from Myo2 in daughter cells. Sequential post-translational modifications of Mmr1, i.e., phosphorylation followed by ubiquitination, are essential for Mmr1 degradation and mitochondrial release from Myo2. Defects in Mmr1 degradation cause stacking and deformation of mitochondria at the bud-tip and bud-neck, where Myo2 accumulates. Compared to wild-type cells, mutant cells with defects in Mmr1 degradation possess an elevated mitochondrial membrane potential and produce higher levels of reactive oxygen species (ROS), along with hypersensitivity to oxidative stress.
Pirates of the haemoglobin
Daniel Akinbosede1, Robert Chizea1 and Stephen A. Hare1,†
Not all treasure is silver and gold; for pathogenic bacteria, iron is the most precious and the most pillaged of metallic elements. Iron is essential for the survival and growth of all life; however free iron is scarce for bacteria inside human hosts. As a mechanism of defence, humans have evolved ways to store iron so as to render it inaccessible for invading pathogens, such as keeping the metal bound to iron-carrying proteins. For bacteria to survive within humans, they must therefore evolve counters to this defence to compete with these proteins for iron binding, or directly steal iron from them. (…)
An ionophore breaks the multi-drug-resistance of Acinetobacter baumannii
David M.P. De Oliveira1 and Mark J. Walker1
Within intensive care units, multi-drug resistant Acinetobacter baumannii outbreaks are a frequent cause of ventilator-associated pneumonia. During the on-going COVID-19 pandemic, patients who receive ventilator support experience a 2-fold increased risk of mortality when they contract a secondary A. baumannii pulmonary infection. In our recent paper (De Oliveira et al. (2022), Mbio, doi: 10.1128/mbio.03517-21), we demonstrate that the 8-hydroxquinoline ionophore, PBT2 breaks the resistance of A. baumannii to tetracycline class antibiotics. In vitro, the combination of PBT2 and zinc with either tetracycline, doxycycline, or tigecycline was shown to be bactericidal against multi-drug-resistant A. baumannii, (…)
Endomembrane remodeling and dynamics in Salmonella infection
Ziyan Fang1 and Stéphane Méresse1
Salmonellae are bacteria that cause moderate to severe infections in humans, depending on the strain and the immune status of the infected host. These pathogens have the particularity of residing in the cells of the infected host. They are usually found in a vacuolar compartment that the bacteria shape with the help of effector proteins. Following invasion of a eukaryotic cell, the bacterial vacuole undergoes maturation characterized by changes in localization, composition and morphology. In particular, membrane tubules stretching over the microtubule cytoskeleton are formed from the bacterial vacuole. Although these tubules do not occur in all infected cells, they are functionally important and promote intracellular replication. This review focuses on the role and significance of membrane compartment remodeling observed in infected cells and the bacterial and host cell pathways involved.
The small bowel microbiome changes significantly with age and aspects of the ageing process
Gabriela Leite1, Mark Pimentel1,2, Gillian M. Barlow1 and Ruchi Mathur1,3
Gut microbiome changes have been associated with human ageing and implicated in age-related diseases including Alzheimer’s disease and Parkinson’s disease. However, studies to date have used stool samples, which do not represent the entire gut. Although more challenging to access, the small intestine plays critical roles in host metabolism and immune function. In this paper (Leite et al. (2021), Cell Reports, doi: 10.1016/j.celrep.2021.109765), we demonstrate significant differences in the small intestinal microbiome in older subjects, (…)
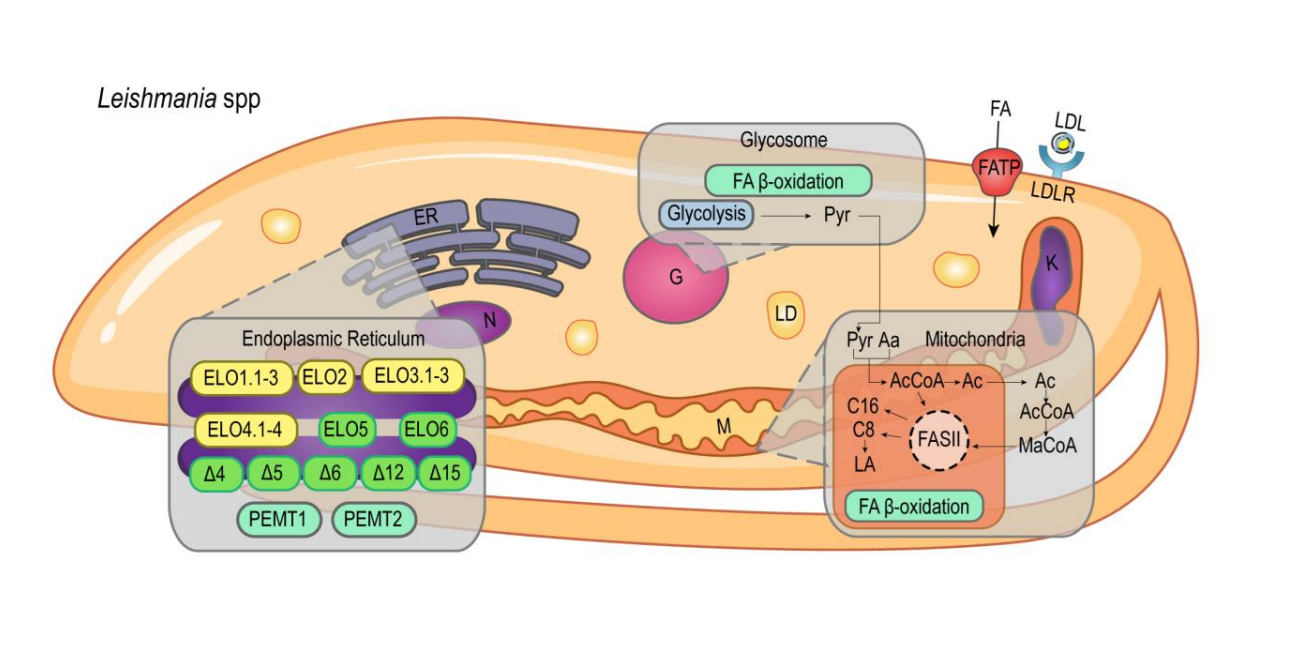
Lipid and fatty acid metabolism in trypanosomatids
Giovana Parreira de Aquino1,#, Marco Antonio Mendes Gomes1,#, Roberto Köpke Salinas2 and Maria Fernanda Laranjeira-Silva1
This work reviews specific aspects of lipid and fatty acid metabolism in the protozoan parasites T. brucei, T. cruzi, and Leishmania spp., as well as the pathways that have been explored for the development of new chemotherapies.
Targeting GATA transcription factors – a novel strategy for anti-aging interventions?
Andreas Zimmermann1, Katharina Kainz1,2, Sebastian J. Hofer1,3, Maria A. Bauer1, Sabrina Schroeder1, Jörn Dengjel4, Federico Pietrocola5, Oliver Kepp6-9, Christoph Ruckenstuhl1, Tobias Eisenberg1,3,10,11, Stephan J. Sigrist12, Frank Madeo1,3,10, Guido Kroemer6-9, 13-15 and Didac Carmona-Gutierrez1
This article comments on work published by Carmona-Gutierrez et al. (Nat Commun., 2019), which identified a natural compound, 4,4′-dimethoxychalcone, inducing autophagy and prolonging lifespan in different organisms through a mechanism that involves GATA transcription factors.
In the beginning was the word: How terminology drives our understanding of endosymbiotic organelles
Miroslav Oborník 1,2
This In the Pit article argues that the naming conventions for biological entities influence research perspectives and methodologies, advocating for mitochondria and plastids to be classified and named as bacteria due to their endosymbiotic origins, with potential implications for our understanding of bacterial prevalence, definitions of the microbiome and multicellularity, and the concept of endosymbiotic domestication.
What’s in a name? How organelles of endosymbiotic origin can be distinguished from endosymbionts
Ansgar Gruber1
This In the Pit article suggests redefining the relationship between hosts and endosymbionts, like mitochondria and plastids, as a single species based on “sexual symbiont integration,” the loss of independent speciation, and congruence in genetic recombination and population sizes, rather than solely on historic classifications or structural properties.
Microbial wars: competition in ecological niches and within the microbiome
Maria A. Bauer1, Katharina Kainz1, Didac Carmona-Gutierrez1 and Frank Madeo1,2
In this Editorial Bauer et al. provide a brief overview on microbial competition and discuss some of its roles and consequences that directly affect humans.
Exploring the mechanism of amebic trogocytosis: the role of amebic lysosomes
Allissia A. Gilmartin1 and William A. Petri, Jr1,2,3
In this article, the authors comment on the study “Inhibition of Amebic Lysosomal Acidification Blocks Amebic Trogocytosis and Cell Killing” by Gilmartin et al. (MBio, 2017), discussing the the role of amebic lysosomes in Trogocytosis, the intracellular transfer of fragments of cell material.
Uncovering the hidden: complexity and strategies for diagnosing latent tuberculosis
Mario Alberto Flores-Valdez
This editorial postulates that advanced proteomic and transcriptomic techniques are evolving and may enhance the detection of latent tuberculosis, thereby distinguishing true M. tuberculosis infections from other conditions, which is vital for controlling potential reactivation and transmission.
The Yin & Yang of Mitochondrial Architecture – Interplay of MICOS and F1Fo-ATP synthase in cristae formation
Heike Rampelt1 and Martin van der Laan2
This Editorial posits that mitochondrial cristae architecture is shaped by the interplay of MICOS and ATP synthase, with a recent study illuminating their roles in cristae formation and maintenance.
When a ribosomal protein grows up – the ribosome assembly path of Rps3
Brigitte Pertschy
This article comments on two papers by Mitterer et al., which followed yeast protein Rps3, highlighting the sophisticated mechanisms for protein protection, nuclear transport, and integration into pre-ribosomal particles for final assembly with 40S subunits.
Microbial Cell
is an open-access, peer-reviewed journal that publishes exceptionally relevant research works that implement the use of unicellular organisms (and multicellular microorganisms) to understand cellular responses to internal and external stimuli and/or human diseases.
you can trust
Can’t find what you’re looking for?
You can browse all our issues and published articles here.
FAQs
Peer-reviewed, open-access research using unicellular organisms (and multicellular microorganisms) to understand cellular responses and human disease.
The journal (founded in 2014) is led by its Editors-in-Chief Frank Madeo, Didac Carmona-Gutierrez, and Guido Kroemer
Microbial Cell has been publishing original scientific literature since 2014, and from the very beginning has been managed by active scientists through an independent Publishing House (Shared science Publishers). The journal was conceived as a platform to acknowledge the importance of unicellular organisms, both as model systems as well as in the biological context of human health and disease.
Ever since, Microbial Cell has very positively developed and strongly grown into a respected journal in the unicellular research community and even beyond. This scientific impact is reflected in the yearly number of citations obtained by articles published in Microbial Cell, as recorded by the Web of Science (Clarivate, formerly Thomson/Reuters):
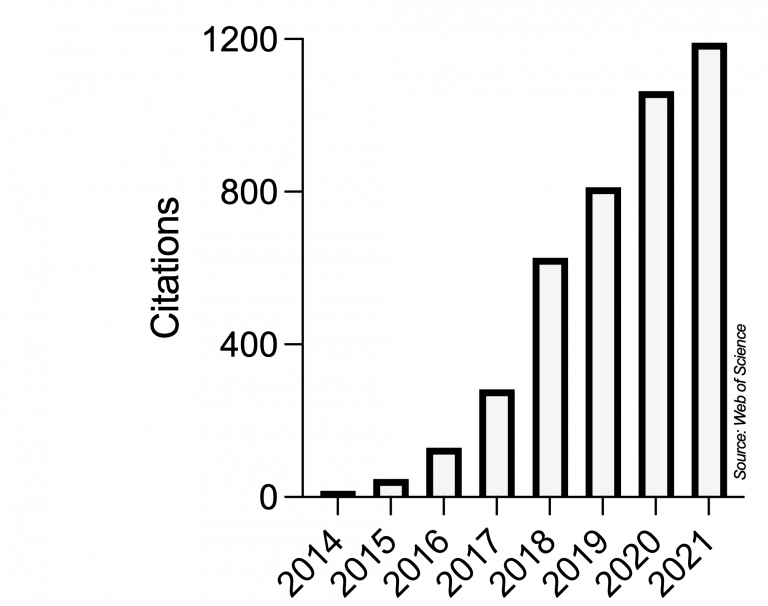
The scientific impact of Microbial Cell is also mirrored in a series of milestones:
2015: Microbial Cell is included in the Emerging Sources Citation Index (ESCI), a selection of developing journals drafted by Clarivate Analytics based on the candidate’s publishing standards, quality, editorial content, and citation data. Note: As an ESCI-selected journal, Microbial Cell is currently being evaluated in a rigorous and long process to determine an inclusion in the Science Citation Index Expanded (SCIE), which allows the official calculation of Clarivate Analytics’ impact factor.
2016: Microbial Cell is awarded the so-called DOAJ Seal by the selective Directory of Open Access Journals (DOAJ). The DOAJ Seal is an exclusive mark of certification for open access journals granted by DOAJ to journals that adhere to outstanding best practice and achieve an extra high and clear commitment to open access and high publishing standards.
2017: Microbial Cell is included in Pubmed Central (PMC), allowing the archiving of all the journal’s articles in PMC and PubMed.
2019: Microbial Cell is indexed in the prestigious abstract and citation database Scopus after a thorough selection process. This also means that Microbial Cell obtains, for the first time, an official Scopus CiteScore as well as an official journal ranking in the Scimago Journal and Country Ranking.
2022: Microbial Cell’s CiteScore reaches a value of 7.2 for the year 2021, positioning Microbial Cell among the top microbiology journals (previously available CiteScores: 2019: 5.4; 2020: 5.1).
2022: Microbial Cell is indexed in the highly selective Science Citation Index Expanded™, which covers approx. 9,500 of the world’s most impactful journals across 178 scientific disciplines. In their journal selection and curation process, Clarivate´s editors apply 24 ‘quality’ criteria and four ‘impact’ criteria to select the most influential journals in their respective fields. This selection is also a pre-requisite for inclusion in the JCR, which features the impact factor.
2022: Microbial Cell is listed in the Journal Citation Reports™ (JCR), and obtains its first official Journal Impact Factor™ (JIF) for the year 2021: 5.316.Check Article Types and Manuscript Preparation guidelines. Submit online via Scholastica.


Sulfur dioxide resistance in Saccharomyces cerevisiae: beyond SSU1
Estéfani García-Ríos1 and José Manuel Guillamón1
This article discusses the importance of understanding sulfite resistance in Saccharomyces cerevisiae due to its use in winemaking and the potential role of the transcription factor Com2. While the SSU1 gene and its activity have been correlated with sulfite tolerance, the work by Lage et al. (2019) indicates that Com2 might control a large percentage of the genes activated by SO2 and contribute to the yeast’s protective response, offering new insights into the molecular factors influencing this oenological trait.